No products in the cart.
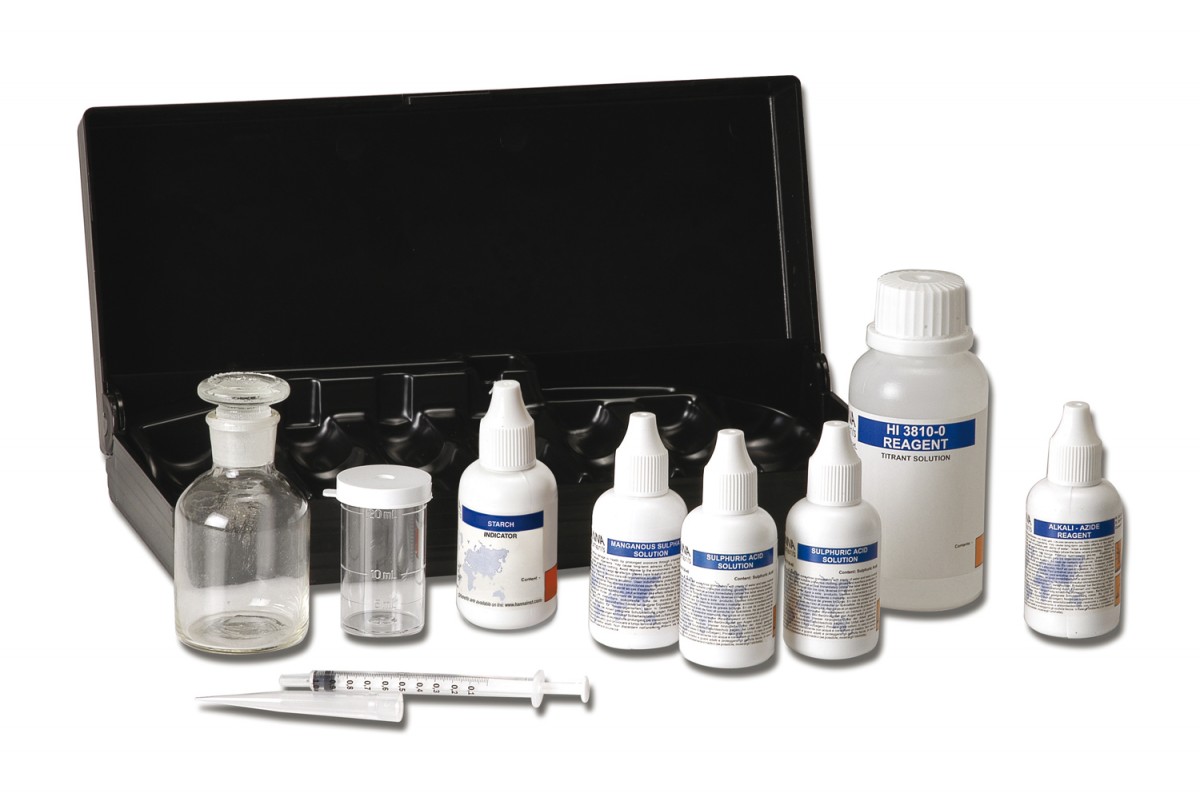
HI3810 CTK Dissolved Oxygen
฿4,494.00
Test kit for the chemical determination of oxygen dissolved in the range 0 to 10.0 mg / l (ppm).
110 tests
28 in stock
The HANNA dissolved oxygen portable test kit can determine the oxygen concentration in water quickly and easily. A modified Winkler method is used. Manganous ions react with oxygen in the presence of potassium hydroxide to form a manganese oxide precipitate. An azide is present to prevent any nitrite ions from interfering with the test. On addition of acid, manganese oxide hydroxide oxidizes the iodide to iodine. Since the amount of iodine generated is equivalent to the oxygen in the sample, the concentration of iodine is calculated by titration of thiosulfate ions that reduce the iodine back to iodide ions.
Order Information:
HI 3810 test kit comes with 30 mL manganous sulfate solution, 30 mL alkali-azide reagent, 60 mL sulfuric acid solution (2), 10 mL starch indicator, 120 mL titrant solution, glass bottle with stopper, 10 mL calibrated vessel and calibrated syringe with tip.
Product Manuals
![]() Manual: Download
Manual: Download
Specification :
| Method | Titration | |
| Range | 0.0-10.0 mg/L | |
| Smallest Increment | 0.1 mg/L | |
| Chemical Method | Modified Winkler | |
| Number of Tests | approx. 110 | |
| Weight | 910 g |
Related products
All Product
฿642.00
Chemischer Testkit zur Bestimmung der Härte im Bereich 0 bis 150 mg/l (ppm). Packungsgröse: 50 Tests
All Product
฿5,885.00
A test kit for the determination of chemical copper in the range 0 to 2.5 mg / l (ppm). 100 Tests
All Product
฿1,337.50
Chemical test kit for the determination of the total chlorine in the range 0 to 2.5 mg / l (ppm). 50 tests
All Product
฿1,712.00
Chemical test kit for the determination of total hardness in the range 0.0 to 30.0 mg / l / 0 to 300 mg / l (ppm). 100 Tests
All Product
฿5,136.00
A test kit for the determination of chemical sulphite in the range 0 to 20 mg / l / 0 to 200 mg / l (ppm). 110 tests
All Product
฿4,815.00
Chemical test kit for the determination of free and total chlorine in the range 0.0-0.70/0.0-3.5 mg / l (ppm). 200 tests
All Product
฿1,337.50
Chemical test kit for the determination of phosphate in the range 0-5 mg / l (ppm). 50 tests
All Product
฿4,280.00
Chemical test kit for the determination of hydrogen peroxide in the range 0.00 to 2.00 mg / L / 0.0 to 10.0 mg / l (ppm). 100 Tests