ไม่มีสินค้าในตะกร้า
HI3887 CTK pH & Free Chlorine
฿2,728.50
Chemical test kit for the determination of pH, free chlorine in Cl2: 0.5 mg / l / pH 6.0 to 8.5.
50/100 tests
มีสินค้าอยู่ 1
รหัสสินค้า: HI3887
หมวดหมู่: ชุดทดสอบเคมี, สินค้าทั้งหมด, แบบจานเทียบสี, แบบเทียบสี, แบบไทเทรต
Brand: HANNA Instruments Inc.
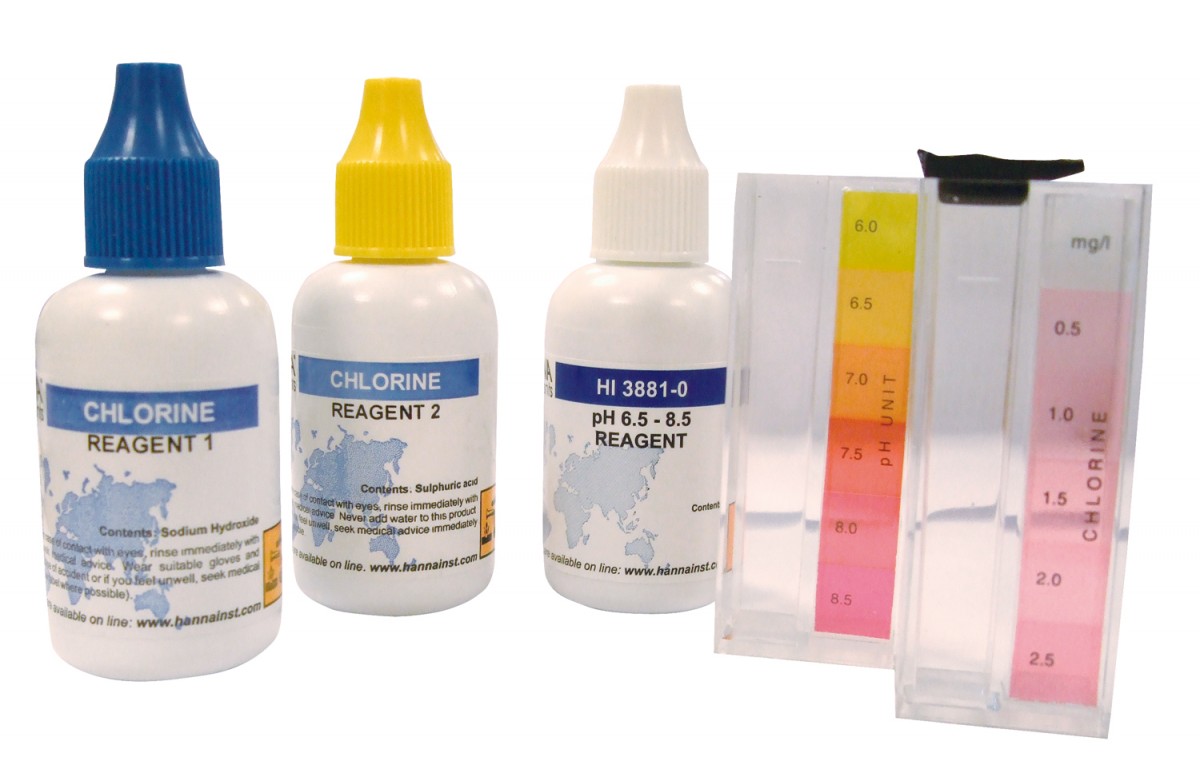
ชุดทดสอบ HI3887 มาพร้อมกับลูกบาศก์เปรียบเทียบสี (2), รีเอเจนต์ 20 มล. 1, รีเอเจนต์ 15 มล. 2, 25 มล. HI 3881-0 รีเอเจนต์ pH 6.0-8.5
คลอรีนอิสระทำปฏิกิริยากับแอมโมเนียมไอออนและสารประกอบอินทรีย์เพื่อสร้างสารประกอบคลอรีน ส่งผลให้ความสามารถในการฆ่าเชื้อลดลงเมื่อเทียบกับคลอรีนอิสระ สารประกอบคลอรีนเหล่านี้ร่วมกับคลอรามีนจะเกิดเป็นคลอรีนรวม คลอรีนรวมและคลอรีนอิสระส่วนเกินรวมกันส่งผลให้มีคลอรีนทั้งหมด แม้ว่าคลอรีนอิสระจะมีศักยภาพในการฆ่าเชื้อสูงกว่ามาก แต่คลอรีนผสมมีความคงตัวสูงกว่ามากและมีความผันผวนน้อยกว่า ควรมีระดับคลอรีนอิสระที่ไม่ทำปฏิกิริยาเพียงพอสำหรับการฆ่าเชื้ออย่างเพียงพอ แต่ไม่มากจนเป็นอันตราย
Free chlorine reacts with ammonium ions and organic compounds to form chlorine compounds resulting in diminished disinfecting capabilities compared with free chlorine. These chlorine compounds together with chloramines form combined chlorine. Combined chlorine and excess free chlorine together result in total chlorine. While free chlorine has a much higher disinfectant potential, combined chlorine has a much higher stability and has a lesser volatility. There should be sufficient levels of unreacted free chlorine for adequate disinfection, but not so much as to become harmful.
Order Information:
HI3887 test kit comes with color comparison cubes (2), 20 mL reagent 1, 15 mL reagent 2, 25 mL HI 3881-0 pH 6.0-8.5 reagent
Specification :
| Method | Colorimetric | |
| Range | Cl²: 0.0-2.5 mg/L pH: 6.0-8.5 pH |
|
| Smallest Increment | Cl²: 0.5 mg/L pH: 0.5 pH |
|
| Chemical Method | DPD pH indicator |
|
| Number of Tests | Cl²: approx. 50 pH: 100 |
|
| Weight | 280 g |
สินค้าที่เกี่ยวข้อง
สินค้าทั้งหมด
฿3,103.00
Range 0-999 ppb | ASTM Method D1687-92 diphenylcarbonate | Resolution 1 ppb
สินค้าทั้งหมด
฿3,103.00
Range 0:00 to 2:00 ppm | ASTM D859 Method | 0.01ppm resolution
สินค้าทั้งหมด
฿3,103.00
Range 0 - 150 ppm | ferrous sulfate method | Resolution 1ppm
สินค้าทั้งหมด
฿12,305.00
ช่วงการวัด 0.00-2.50ppm
สินค้าทั้งหมด
฿3,103.00
Range 0 - 600 ppb NO2-N | EPA method 354.1 | Resolution 1 ppb
สินค้าทั้งหมด
฿3,103.00
Range 0:00 to 2:50 ppm | ascorbic acid method | Resolution 0.01ppm